Report

Резюме
- Disclosed deal values surpassed previous highs to reach $40.7 billion in 2019, with the Nestlé Skin Health acquisition composing about 25% of the total.
- Biopharma buyout activity was slightly up, to 85 deals in 2019 from 79 in 2018.
- Products, services and HCIT supporting R&D and commercialization efforts of drugmakers continued to attract the highest interest, but early-stage technology investment also grew.
- Within services, clinical contract research organizations and contract development and manufacturing organizations were hot, as well as other emerging or niche service lines such as cell and gene therapy, institutional review boards and commercialization.
This article is part of Bain’s 2020 Global Healthcare Private Equity and Corporate M&A Report. Explore the contents of the report here or download the PDF to read the full report.
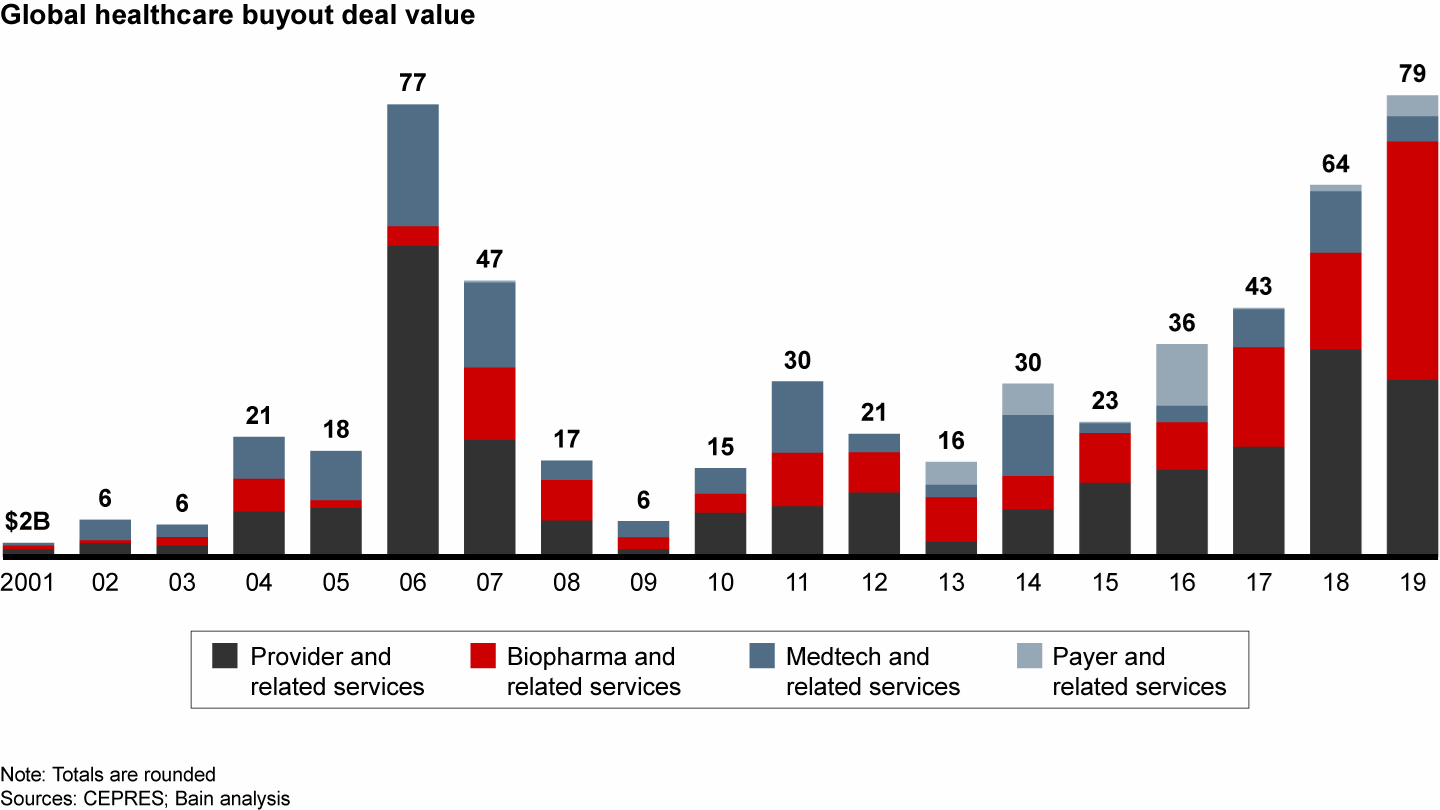
Disclosed deal value reached the highest level ever in 2019

Both private equity and corporate buyers showed strong interest in biopharma throughout 2019. PE investors like the attractive absolute returns, while corporates leverage M&A to supplement their own portfolios with new assets and capabilities.
Their enthusiasm registers despite the political and regulatory uncertainties across the globe that could affect drug pricing. In early 2020, pharma companies continue to take price on their branded products in the US (in line with 2019 rises), implying limited pressure on price, at least before the presidential and congressional election in November.
Deal volume increased slightly to 85 deals in 2019, compared with 79 in 2018, but total disclosed value more than doubled from $16.5 billion to a record high $40.7 billion. The largest deal of the year in healthcare, EQT and ADIA ’s $10.1 billion carve-out acquisition of Nestlé Skin Health’s business, accounted for about 25% of the value. Overall, the attractive growth profile of the sector and intense competition for deals drove valuations to record levels.
Corporate interest in biopharma services may heighten competition, but it also offers PE sponsors a route to exit at the end of the holding period as corporate buyers have shown a propensity to make large investments. 2019 saw striking examples of this in the gene therapy CDMO space: Thermo Fisher Scientific bought Brammer Bio from Ampersand Capital Partners for $1.7 billion, and Catalent acquired Paragon Biosciences from Camden Partners and NewSpring Capital for $1.2 billion. PE Hub reported that the sellers achieved a 31 times multiple on invested capital (MOIC) on the Paragon sale.
Brisk activity worldwide
From a regional standpoint, investment activity split fairly evenly during the year. North America accounted for 36% of deal activity and 42% of disclosed deal value, and Europe represented 28% of activity and 41% of deal value, largely driven by the Nestlé Skin Health acquisition.
Investors in Europe have become more sophisticated over the years. They started with healthcare-light assets, then expanded to over-the-counter and mature drugs. Recently, they expanded into healthcare-heavy territory, addressing the full reimbursement and cost containment risks. The most sophisticated investors have worked in specialized areas requiring deeper experience to articulate a relevant thesis and develop the assets.
At the same time, branded generic platforms have continued to be attractive for PE investors, though investments in STADA Arzneimittel, Zentiva and Recordati have raised the bar for new PE investors to invest in European generics as these three platforms (and other similar ones, such as Karo Pharma in Scandinavia) can offer higher valuations to sellers. For the next round, investors must bring more money or more sophistication, or both.
In the Asia-Pacific region, biopharma continued its long-term growth trajectory. The region accounted for 33% of deal volume in 2019 and 16% of value. Most of the capital fed smaller, early-stage companies. As with other healthcare sectors in the region, biopharma assets appeal to investors due to the positive underlying macro fundamentals, including rising incomes and aging populations. In fact, the region’s largest deal of the year was Centurium Capital’s $1.9 billion take-private offer for the remainder of China Biologic Products Holdings, a Chinese plasma collection network and fractionator.
Biopharma held steady in China, where the government is pushing hard to reduce the country’s dependence on multinational corporations for prescription medications through its bulk-buying program. Such regulatory shifts have opened the door for Chinese pharma companies to win large hospital and other supplier contracts. As the effects of regulation settle and near-term uncertainty clears, deal activity should increase.
Across the regions, four major investment themes emerged or progressed during the year:
- branded consumer pharmaceuticals, in areas with limited pricing pressure and defendable niches;
- biopharma services such as contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs), including newer areas such as cell and gene therapy and commercialization services;
- HCIT that improves efficiency of pharma companies across the value chain, with a specific focus on drug development; and
- broader life science investments, including earlier stage investments carrying clinical and technical risk.
1. Branded consumer pharmaceuticals
PE interest in branded biopharmaceuticals remained strong in areas with low R&D risk. Investors sought areas with limited pricing pressures and defendable niches in therapeutic areas such as dermatology, ophthalmology, central nervous system and in vitro fertilization, as well as in countries where branding still has clout. This was doubly true for consumer-heavy therapeutic areas such as aesthetics and skin care, due to the lower exposure to pricing pressure. These assets also require less R&D spending, have fewer regulatory complexities than patented drugs, face less competitive pressure and involve more consumer branding, which is easier to execute during the holding period.
The largest healthcare deal of the year came in dermatology as EQT and ADIA acquired Galderma, the business unit formerly known as Nestlé Skin Health, which produces branded dermatology drugs and products, for $10.1 billion. The deal was premised on the strong underlying growth of the aesthetics business in many countries (as demonstrated by market leader Allergan), a promising dermatology portfolio, the relative ease to carve out the asset, and room to improve performance.
2. Biopharma services
Investors continued their keen interest in services, with 51% of deals and 55% of deal values in biopharma going to services and HCIT. This is due to the ability to participate without significant clinical or end-market pricing risk, and capitalize on the trend of biopharma companies continuing to outsource noncore activities as well as pursuing efficiency and cost improvements.
CMOs and CROs continued to garner high interest and accounted for 32% of biopharma deals during 2019. For example, Permira acquired Cambrex Corporation, a small molecule CDMO, for $2.4 billion. Permira also bought Quotient Sciences, a provider of contract research and drug development services.
The cell and gene therapy market continues to ride high, including the approval of Zolgensma to treat spinal muscular atrophy in 2019, and makes up roughly 10% of pharma’s late stage pipelines. This technology requires different R&D, manufacturing and commercialization models from traditional biopharma. In 2019, the cell and gene therapy CDMO market drew significant capital from PE sponsors and corporates. EQT bought Aldevron, a producer of plasmids. Thermo Fisher purchased Brammer Bio for $1.7 billion and Catalent purchased Paragon Bioservices.
In addition, there was significant interest in other pharma services offerings across the value chain, including institutional review boards (IRBs), regulatory affairs and commercialization services such as medical affairs, pricing, hub services and market access. The niche IRB market drew particular interest last year, with the sale of two leading competitors, WIRB-Copernicus Group and Advarra.
Commercialization services gained ground as products narrow to fit a niche and a generic approach to commercialization no longer works. Smaller pharma firms often lack in-house commercialization capabilities and must look for help outside across a variety of services lines.
For instance, Publicis Healthcare Solutions, specializing in remote engagement, field outsource solutions, and clinical and medical solutions, was acquired by Altamont Capital Partners and renamed Amplity Health. EVERSANA, backed by Water Street, continued to build out its rare disease platform and acquired Cornerstone Research Group, a provider of HEOR services, Alamo, a provider of field sales, marketing and clinical solutions for biopharma, and BexR Logistix Telesales, a third-party logistics company.
3. HCIT that supports drug development through commercialization
HCIT drew interest as well, especially for assets that support drugmakers in developing, manufacturing and commercializing new therapies. Drug development is expensive and subject to pricing pressure, so pharma companies are trying to improve both speed to market and cost. Data analytics can help support clinical trials as well as use virtual trials to replace or enhance traditional clinical trials. For example, Water Street Healthcare Partners and JLL Partners acquired THREAD Research, a provider of virtual research technology.
4. Early-stage life sciences technologies
Early-stage life science investing fits a set of buyout funds that have an appetite for development and technology risk and possess specific capabilities to source and evaluate opportunities. This continued to be an investment theme in 2019 as specific funds put capital to work in this segment.
Bain Capital raised $900 million for its second fund dedicated to life sciences. Silverlake put $1 billion into Verily Life Sciences, the Alphabet subsidiary, which is a life sciences technology company focused on partnerships with pharma, life sciences and medtech companies. KKR co-led investment in BridgeBio Pharma’s latest private funding round in January, before its June IPO.
Investors also showed greater interest in tools that support life sciences. For example, GTCR invested in Cole-Parmer, a specialty lab equipment provider, two years after selling the company to Golden Gate.
How to win despite high valuations
We expect the biopharma sector to maintain its appeal for investors. As an indicator of broader demand, venture-capital investments in therapeutics remain strong, albeit deals skew to smaller and more risky, earlier-stage bets. There are multiple hunting grounds for investment opportunities across the value chain, but a few specific areas of interest may offer high value for investors in the coming year.
Services and HCIT across the value chain will remain the most fruitful territory, and pricing pressure will amplify the need for efficient drug development and commercial effectiveness. This territory includes niches that provide opportunities for building platforms that address specific customer needs (smaller biopharma) or therapeutic area expertise (oncology or rare disease). Pharma has pain points along its value chain, for example in site and patient recruitment, where newer services and HCIT offerings could improve efficiency.
In addition, pharma’s development model is using more real-world data and evidence, and pharma needs help with generating and prosecuting that data and evidence for such activities as virtual trials and lead optimization.
Branded pharma in particular will attract investors. Besides the existing niches for private equity, this may expand to include areas that have less interest to corporates, such as anti-infective therapies where the commercial business model needs an overhaul. Generics may start to provide an attractive countercyclical investment strategy; while they have not been an active investment area in the US recently, there are signs that the big price declines of recent years are abating and that the industry’s structure will stabilize.
This article is part of Bain’s 2020 Global Healthcare Private Equity and Corporate M&A Report. Explore the contents of the report here or download the PDF to read the full report.